Not long ago, the only way to tell whether a patient with dementia had Alzheimer’s disease was to do an autopsy for the presence of amyloid plaque and other signs of degeneration in the brain. In recent years, new tests can detect the presence of amyloid, a telltale protein of Alzheimer’s, and other biological signs long before the onset of symptoms. Soon, doctors may routinely make definitive diagnoses of Alzheimer’s with a simple blood test, even before symptoms of dementia become apparent.
很久以前,判断痴呆症患者是否患有阿尔茨海默病的唯一方法是进行尸检,检查大脑中是否存在淀粉样斑块和其他退化迹象。近年来,新的检测方法可以在症状出现之前很长时间就检测出淀粉样蛋白(一种阿尔茨海默氏症的标志性蛋白质)的存在以及其他生物迹象。不久的将来,医生甚至可以在痴呆症状显现之前,通过简单的血液化验就对阿尔茨海默氏症做出明确诊断。
An early diagnosis of Alzheimer’s is not worth much if there’s nothing you can do about it. But new effective treatments that slow the progress of the disease have become available: the drug lecanemab, recently approved by the FDA, and a new one called donanemab, which slowed cognitive decline in trials. The availability of effective treatments, together with technologies for detecting Alzheimer’s in the early stages, when those treatments can be most effective, have radically changed the outlook for Alzheimer’s patients and their loved ones. The notion of attacking Alzheimer’s in the brain before clinical symptoms emerge, long merely an aspiration, is starting to look like a practical strategy.
如果对阿尔茨海默氏症束手无策,那么早期诊断的价值就不大了。但是,减缓疾病进展的新的有效治疗方法已经问世:最近获得美国食品及药物管理局批准的药物 lecanemab 和一种名为 Donanemab 的新药在试验中减缓了认知能力的下降。有效治疗方法的出现,加上在治疗效果最佳的早期阶段检测阿尔茨海默氏症的技术,从根本上改变了阿尔茨海默氏症患者及其亲人的前景。长期以来,在临床症状出现之前对大脑中的阿尔茨海默氏症进行治疗一直只是一种愿望,而现在这种想法已开始成为一种切实可行的策略。
Advances in early detection and treatment come as welcome news, but they imply a looming public-health challenge. Being able to screen for Alzheimer’s and administer treatments before symptoms arise would vastly increase the number of people who need attention. Public-health institutions are almost universally inadequate for the task. There are large disparities in the impact of Alzheimer’s and in access to care in the U.S. and around the world. Pilot programs in communities around the world are showing how it might be done.
在早期检测和治疗方面取得的进展令人欣喜,但这也意味着一个迫在眉睫的公共卫生挑战。如果能够在症状出现之前筛查阿尔茨海默氏症并进行治疗,那么需要关注的人数将大大增加。公共卫生机构几乎普遍无法胜任这项任务。在美国和世界各地,阿尔茨海默氏症的影响和获得治疗的机会存在巨大差异。世界各地社区的试点项目正在展示如何做到这一点。
Meanwhile, the new optimism rippling through the research field is palpable. “Having been in this field for 20 years, the idea that I can finally offer treatments that biologically slow the disease is incredibly exciting,” says Gil Rabinovici, who directs the Alzheimer’s Disease Research Center at the University of California, San Francisco. “There’s a lot more work to do, but the feeling is that our understanding and ability to measure and treat the disease is coming together in a new way.”
与此同时,新的乐观情绪也在研究领域悄然蔓延。"加利福尼亚大学旧金山分校阿尔茨海默病研究中心主任吉尔-拉比诺维奇说:"我在这个领域已经工作了 20 年,终于可以提供从生物学角度减缓疾病的治疗方法了,这种想法令人无比兴奋。"还有很多工作要做,但我们的感觉是,我们对这种疾病的理解以及测量和治疗这种疾病的能力正在以一种新的方式结合在一起。
An enormous toll 巨大损失
About one in nine Americans over 65 have Alzheimer’s disease, according to figures from the Alzheimer’s Association. The numbers are higher for several segments of the population, including women, Black Americans and Hispanics. The number of people with Alzheimer’s is expected to more than double in 25 years.
根据阿尔茨海默氏症协会(Alzheimer's Association)的数据,65 岁以上的美国人中,约九分之一患有阿尔茨海默氏症。包括妇女、美国黑人和西班牙裔在内的一些人群的患病率更高。预计在 25 年内,阿尔茨海默氏症患者的人数将增加一倍以上。
It is a cruel, relentless disease. “It progressively robs you of who you are,” says neuroscientist Donna Wilcock, director of Indiana University’s Center for Neurodegenerative Disorders. Families carry much of the weight, she adds. The annual cost of Alzheimer’s care in the U.S. has reached $345 billion, the Alzheimer’s Association estimates—and that doesn’t count the $340 billion worth of unpaid care put in by an estimated 11 million family members and other caregivers of U.S. Alzheimer’s patients in 2022. Other estimates run even higher (see “The Ten Trillion Dollar Disease,” on page 24).
这是一种残酷无情的疾病。"印第安纳大学神经退行性疾病中心主任、神经科学家唐娜-威尔科克(Donna Wilcock)说:"它会逐渐剥夺你的自我。她补充说,家庭承担了大部分重担。据阿尔茨海默氏症协会估计,美国每年用于阿尔茨海默氏症护理的费用已高达 3450 亿美元,这还不包括 2022 年美国约 1100 万阿尔茨海默氏症患者的家庭成员和其他护理人员所付出的价值 3400 亿美元的无偿护理费用。其他估计数字甚至更高(请参阅第 24 页 "十万亿美元的疾病")。
Modern medicine has made enormous strides in treating cancer, diabetes, heart disease, and even other neurodegenerative diseases such as Parkinson’s and multiple sclerosis. But for years, everything medicine could throw at Alzheimer’s seemed to bounce off. The main research strategy has been to try to come up with drugs that attack the plaque that for more than a century has been known to be present in the brain tissue of deceased Alzheimer’s patients. But the dozens of experimental drugs that reduced brain plaque in mice with Alzheimer’s-like symptoms failed to make any detectable difference in cognitive decline in human drug trials.
现代医学在治疗癌症、糖尿病、心脏病,甚至帕金森氏症和多发性硬化症等其他神经退行性疾病方面取得了巨大进步。但多年来,医学界对阿尔茨海默氏症所做的一切似乎都是徒劳的。一个多世纪以来,人们一直知道阿尔茨海默氏症患者的脑组织中存在斑块,而主要的研究策略就是试图研制出能够攻击这种斑块的药物。但是,在人类药物试验中,数十种能够减少具有类似阿尔茨海默氏症症状的小鼠脑斑块的实验性药物未能对认知能力下降产生任何可察觉的影响。
Then, in 2021, a leap of progress came with the Food and Drug Administration approval of aducanumab, an amyloid-beta-attacking monoclonal antibody—a lab-made version of an antibody found in the human immune system. Aducanumab was the first drug ever approved for slowing cognitive decline in Alzheimer’s patients. But it was a controversial decision, in part because trial data showed at best hints of a possible small average slowing in cognitive decline.
2021 年,美国食品药品管理局批准了阿杜卡单抗,这是一种攻击淀粉样蛋白-β 的单克隆抗体--一种在人类免疫系统中发现的抗体的实验室制造版本,从而实现了飞跃性的进步。阿杜单抗是有史以来第一种获准用于减缓阿尔茨海默病患者认知能力衰退的药物。但这是一个有争议的决定,部分原因是试验数据显示,这种药物最多只能平均减缓一小部分认知能力的衰退。
Much of that uncertainty evaporated in January 2023, when the FDA approved a second plaque-fighting monoclonal antibody, lecanemab. This time, the regulators saw clear evidence that on average the drug slowed the cognitive decline in patients with early symptoms of Alzheimer’s. And that thrilling news was still reverberating through the research and patient communities when data released in July 2023 from a phase 3 trial of a third monoclonal antibody, donanemab, showed even better results, slowing the decline in memory and thinking by 35 percent compared to placebo. Almost half the study subjects who received donanemab showed no worsening of symptoms during a year of treatment, compared to only 29 percent in the placebo group.
2023 年 1 月,美国食品和药物管理局批准了第二种对抗斑块的单克隆抗体--莱卡奈单抗(lecanemab),这在很大程度上消除了不确定性。这一次,监管机构看到了明确的证据,即平均而言,这种药物减缓了阿尔茨海默氏症早期症状患者认知能力的下降。2023 年 7 月,第三种单克隆抗体多奈单抗的三期试验数据公布,与安慰剂相比,多奈单抗能将记忆力和思维能力下降的速度减缓 35%,这一令人振奋的消息仍在研究界和患者群体中回荡。在接受多那尼单抗治疗的研究对象中,近一半在一年的治疗期间症状没有恶化,而安慰剂组只有 29%。
“This is one of the things that patients and families say is important to them—to stay at the same level longer to keep doing what they enjoy doing,” says Mark Mintun, who heads neuroscience research and development at Eli Lilly, the pharmaceutical company that developed donanemab.
"开发多那尼单抗的制药公司礼来公司负责神经科学研发的马克-明顿(Mark Mintun)说:"这是病人和家属认为对他们很重要的事情之一--在同一水平上保持更长时间,继续做他们喜欢做的事情。
The sudden appearance of helpful treatments has been revitalizing for both the patient and research communities, says Indiana University’s Wilcock. “Patients and families had lost a lot of hope, and this restores it,” she says. “I truly believe it’s the beginning of the successful treatment of the disease.”
印第安纳大学的威尔科克说,有益疗法的突然出现给患者和研究界都带来了新的活力。"她说:"患者和家属失去了很多希望,而这让他们重新燃起了希望。"我真的相信这是成功治疗这种疾病的开始。
Continually better drugs are likely to follow in a steady stream over the coming years and decades, experts say. Alzheimer’s research may be similar to multiple sclerosis, another complex neurological disease. The first approved drugs in the 1990s were about 30 percent effective, and now several drugs are more than 80 percent effective.
专家们说,在未来几年和几十年里,更好的药物可能会源源不断地出现。阿尔茨海默病的研究可能与另一种复杂的神经系统疾病多发性硬化症类似。20 世纪 90 年代首批获批的药物有效率约为 30%,而现在有几种药物的有效率超过了 80%。
AXS Biomedical Animation Studio
AXS 生物医学动画工作室
Diagnostics breakthroughs
诊断突破
Several factors have led to the sudden emergence of effective drugs. One is that the newer drugs are aiming at the right kind of amyloid plaque. There are many types, most emerging from variations of the amyloid beta peptide, but research has gradually pointed at particular forms, giving drug developers more specific and fruitful targets. A second factor is the improving ability to produce monoclonal antibodies that are more effective at hitting their targets.
有几个因素导致了有效药物的突然出现。其一是新药瞄准了正确类型的淀粉样蛋白斑块。淀粉样蛋白斑块有很多种类型,大多数来自于淀粉样蛋白 beta 肽的变异,但研究已逐渐指向特定的形式,从而为药物开发人员提供了更具体、更富有成效的靶点。第二个因素是生产单克隆抗体的能力不断提高,这种抗体能更有效地击中目标。
But what may be the biggest single factor in the breakthrough treatment progress isn’t really about the treatments themselves. Rather, it’s better diagnosis.
但是,治疗取得突破性进展的最大因素其实并不在于治疗本身。而是更好的诊断。
A positron emission tomography, or PET, scan, involves injecting a radioactive tracer compound into a patient and then scanning for photons emitted. If the tracer binds mostly to something in the body associated with a particular disease, then the scan can be useful in diagnosis. PET scans capable of detecting amyloid beta plaque in living human patients have been around since 2004, but the early versions required massive machines that cost more than a million dollars to install, and even then the results didn’t give a reliable diagnosis of Alzheimer’s disease. It wasn’t until 2017 that affordable PET scans became capable of reliable plaque-based Alzheimer’s disease diagnosis.
正电子发射断层扫描(PET)是将放射性示踪化合物注入患者体内,然后扫描其发射的光子。如果示踪剂主要与体内某种与特定疾病相关的物质结合,那么扫描就可以用于诊断。能够检测活人体内淀粉样蛋白 beta 斑块的 PET 扫描早在 2004 年就出现了,但早期版本需要安装耗资超过 100 万美元的大型机器,即便如此,扫描结果也不能可靠地诊断出阿尔茨海默氏症。直到 2017 年,价格低廉的 PET 扫描才能够进行可靠的基于斑块的阿尔茨海默病诊断。
When clinicians and researchers started accumulating results from those scans, they discovered two things. First, as many of 30 percent of people diagnosed with Alzheimer’s disease based on cognitive assessments didn’t have high levels of the plaque, which meant they had a different type of dementia and had been misdiagnosed. Thus, some of the plaque-reducing drugs that had failed in earlier trials probably hadn’t done as well as they might have because they included patients who had a different condition that had little to do with plaque.
当临床医生和研究人员开始积累这些扫描结果时,他们发现了两件事。首先,在根据认知评估诊断出患有阿尔茨海默病的患者中,有多达 30% 的人体内的斑块含量并不高,这意味着他们患有不同类型的痴呆症,并且被误诊了。因此,一些在早期试验中失败的减少斑块的药物可能并没有取得预期的效果,因为其中包括了患有与斑块关系不大的不同病症的患者。
Second, PET scan data clearly demonstrated that people with no clinical symptoms sometimes had high levels of plaque, and those people would usually go on to develop symptoms years later. In other words, people with high levels of plaque but no signs of cognitive impairment were simply earlier in the disease progression.
其次,PET 扫描数据清楚地表明,没有临床症状的人有时会有高水平的斑块,而这些人通常会在几年后出现症状。换句话说,斑块含量高但没有认知障碍症状的人只是在疾病进展的早期出现了症状。
Researchers had long believed that the biological seeds of Alzheimer’s are planted long before—even decades before—cognitive impairment is clearly diagnosable, but they had no way of knowing which people were on that path. With PET scans closing that gap, drug trials could include patients positively diagnosed, through detection of plaque, while in the earlier stages of the disease, when a plaque-clearing drug might have a better chance of heading off damage. What’s more, ongoing PET scans can quickly reveal whether a trial drug is actually reducing brain plaque, making it easier to focus on drugs that had the best chance of working. Perhaps most important, these scans can reliably identify patients who have the disease in order to know which ones will likely benefit from approved treatments.
长期以来,研究人员一直认为,阿尔茨海默氏症的生物学种子早在认知障碍被明确诊断之前就已经埋下,甚至早在几十年前就已经埋下,但他们却无从得知哪些患者正处于这一阶段。PET 扫描缩小了这一差距,药物试验可以将通过检测斑块确诊的患者纳入其中,而这些患者正处于疾病的早期阶段,此时清除斑块的药物可能更有机会避免损害。更重要的是,持续的 PET 扫描可以迅速揭示试验药物是否真的能减少脑斑块,从而更容易将重点放在最有可能发挥作用的药物上。也许最重要的是,这些扫描可以可靠地确定哪些患者患有这种疾病,从而知道哪些患者可能会受益于已获批准的治疗方法。
All told, the ability to diagnose the disease more surely and earlier, and to track its biological progression, has helped revolutionize the effort to develop and prescribe treatments.
总而言之,能够更准确、更早地诊断出这种疾病并跟踪其生物学进展,有助于彻底改变治疗方法的开发和处方。
“We don’t have to wait for someone to have symptoms to know if they have cancer,” says UCSF’s Rabinovici. “But 100 years after the initial Alzheimer’s case reports, we were still diagnosing by clinical symptoms confirmed at autopsy. These scans represent the modernization of Alzheimer’s care.”
"加州大学旧金山分校的拉比诺维奇说:"我们不必等到某人出现症状时才知道他是否患了癌症。"但在最初的阿尔茨海默氏症病例报告发布 100 年后,我们仍在通过尸检确认的临床症状进行诊断。这些扫描代表了阿尔茨海默氏症护理的现代化。
Even better, blood tests are emerging that can pick up biomarkers correlated with the buildup of amyloid plaque in the brain, at much lower cost and higher convenience than a PET scan or a spinal tap, the other way of detecting signs of plaque. Blood tests could eventually become a routine screen for people who are in their 50s, when someone on track to experience cognitive decline from Alzheimer’s in their 70s would probably already have detectable levels of plaque.
更妙的是,血液检测可以检测出与大脑中淀粉样蛋白斑块堆积相关的生物标志物,与正电子发射计算机断层扫描或脊髓穿刺(检测斑块迹象的另一种方法)相比,成本更低,更方便。血液检测最终可能成为 50 多岁人群的常规筛查方法,而 70 多岁的阿尔茨海默氏症患者很可能已经检测到了斑块的水平。
Better, less expensive and more convenient diagnostic tests also mean people could more easily get a shot at one of the new plaque-clearing drugs when they may be able to do the most good. Research has shown that patients with mild cognitive decline do better when treated in early stage of the disease. In presymptomatic patients—that is, those who show no cognitive decline, but test positive for plaque—it’s still unproven whether treating them with anti-plaque drugs is effective in slowing or preventing the disease. “It’s an incredibly important question,” says Madhav Thambisetty, chief of the clinical and translational neuroscience section at the National Institute on Aging (NIA) and a neurologist at Johns Hopkins University.
更好、更便宜、更方便的诊断测试也意味着人们可以更容易地获得一种新的清除斑块的药物,因为这些药物可能会发挥最大的作用。研究表明,轻度认知功能衰退患者在疾病早期接受治疗的效果更好。对于无症状的患者,即认知能力没有下降但斑块检测呈阳性的患者,用抗斑块药物治疗是否能有效减缓或预防疾病,目前仍未得到证实。"美国国家老龄化研究所(NIA)临床与转化神经科学部主任、约翰霍普金斯大学神经学家马达夫-坦比塞提(Madhav Thambisetty)说:"这是一个非常重要的问题。
Two ongoing phase 3 drug trials intend to answer the question. The AHEAD 3-45 trial is testing the impact of lecanemab on the onset of clinical symptoms in people who currently have plaque but no symptoms; the TRAILBLAZER-ALZ 3 trial is doing the same with donanemab. Both trials are planned to run through 2027.
目前正在进行的两项三期药物试验打算回答这个问题。AHEAD 3-45 试验正在测试莱卡奈单抗对目前有斑块但无症状的患者出现临床症状的影响;TRAILBLAZER-ALZ 3 试验正在对多那奈单抗进行同样的测试。这两项试验计划持续到2027年。
Genetic testing, too, will be part of the diagnostics picture for Alzheimer’s. Patients can already be tested for the APOE4 gene. About one out of four people have one copy of the gene, which more than doubles the chances of developing Alzheimer’s, and one in 40 or so people have two copies, leading to roughly a 10-fold increase in risk. Researchers have also found APOE4 variations that appear to confer a lower risk of the disease. Over time, other genetic predispositions or protections from the disease are likely to emerge, and some may also lead to new drug targets and therapies.
基因检测也将成为阿尔茨海默氏症诊断的一部分。患者已经可以接受 APOE4 基因检测。大约每四个人中就有一个人拥有该基因的一个拷贝,患阿尔茨海默氏症的几率会增加一倍多,而每 40 人中就有一个人拥有两个拷贝,导致患病风险增加大约 10 倍。研究人员还发现,APOE4 变异似乎会降低患病风险。随着时间的推移,很可能会出现其他的遗传倾向或对疾病的保护作用,其中一些还可能导致新的药物靶点和疗法。
Some researchers also point out that the hunt for ever-earlier signs of impending cognitive decline from Alzheimer’s, while a boon to research and early treatment, requires consideration of privacy and medical ethics issues. Other concerns with genetic and other testing for very early Alzheimer’s include the risk that the results could impact eligibility for employment, life insurance and long-term care.
一些研究人员还指出,寻找阿尔茨海默氏症认知能力即将衰退的早期征兆虽然有利于研究和早期治疗,但需要考虑隐私和医学伦理问题。对极早老年痴呆症进行基因检测和其他检测的其他顾虑还包括,检测结果可能会影响就业、人寿保险和长期护理的资格。
Attacking the disease 对症下药
The drug treatments that attack plaque are going to become increasingly effective, insist most researchers. Now that two anti-plaque drugs have been proven to slow the progress of cognitive decline, researchers and pharmaceutical companies can focus on new, better ways of hitting plaque. That may include new anti-plaque monoclonal antibodies that do a better job. Or it may be pills that work about as well but, unlike most antibodies, don’t need to be ‘infused’ into patients—a biweekly or monthly process in which the drug is slowly dribbled into the body through an intravenous tube, usually in a clinic setting, or sometimes at home with the help of a trained caregiver.
大多数研究人员坚持认为,治疗斑块的药物将越来越有效。现在,两种抗斑块药物已被证明可以减缓认知能力衰退的进程,研究人员和制药公司可以把重点放在新的、更好的治疗斑块的方法上。这可能包括效果更好的新型抗斑块单克隆抗体。或者是效果差不多的药片,但与大多数抗体不同的是,这种药片不需要 "输注 "到患者体内--通常是在诊所,有时是在家里,在训练有素的护理人员的帮助下,每两周或每月通过静脉注射管将药物缓慢滴入患者体内。
Eli Lilly, for example, is developing a monoclonal antibody called remternetug that in early trials appears effective against plaque and may also be easily injectable under the skin like a flu shot.
例如,礼来公司正在开发一种名为 remternetug 的单克隆抗体,在早期试验中,这种抗体似乎对斑块有效,而且还可以像流感疫苗一样方便地皮下注射。
Whether through existing anti-plaque drugs or new ones, researchers expect to see more patients get more benefits as research and clinical experience help find ways to match the right drug to the right patients, and to zero in on the optimal doses and timing.
无论是通过现有的抗斑块药物还是新药,研究人员都希望看到更多的患者能从中获益,因为研究和临床经验有助于找到将正确的药物与正确的患者相匹配的方法,并找到最佳剂量和时机。
If there is a cloud hanging over the prospects for anti-plaque treatments, it is amyloid-related imaging abnormalities, or ARIA. ARIA are essentially small bleeds and minor swelling in the brain, first noticed more than a decade ago in MRI scans of patients who were taking some of the first experimental anti-plaque monoclonal antibodies (and also in some patients who were taking placebo). ARIA have continued to show up in a significant portion of patients in trials for these drugs. In most cases there are no symptoms or only mild ones such as headaches that usually clear up within a few months, but there have sometimes been serious events such as seizures, strokelike symptoms and even death. For that reason, lecanemab carries an FDA “black box” warning, reserved for only drugs with the most serious concerns.
如果说抗斑块治疗的前景笼罩着一层阴云,那就是淀粉样蛋白相关成像异常,或称 ARIA。ARIA 基本上是大脑中的小出血点和轻微肿胀,十多年前首次在服用一些首批试验性抗斑块单克隆抗体的患者(以及一些服用安慰剂的患者)的核磁共振扫描中被发现。在这些药物的试验中,有相当一部分患者继续出现 ARIA。在大多数情况下,患者没有症状或仅有头痛等轻微症状,通常会在几个月内痊愈,但有时也会出现癫痫发作、中风样症状甚至死亡等严重事件。因此,莱卡尼单抗受到了美国食品及药物管理局的 "黑框 "警告,只有存在最严重问题的药物才会受到这种警告。
Few researchers suggest that worries over ARIA merit halting the use of anti-plaque drugs, given how welcome the progress in treatment has been to the Alzheimer’s community and how dire the consequences of leaving the disease untreated. In addition, clinical trials are conducted on elderly patients with many comorbidities that can obscure the cause of symptoms. Still, many are calling for more research. “We have to try to understand the underlying mechanisms of ARIA,” says Wilcock. “And we’re in desperate need of biomarkers that would help us identify the patients at highest risk.” Ultimately, she says, it should be possible to identify each individual’s chances of being helped by an anti-plaque drug, along with the risk of being harmed, so that doctors, patients and families can make informed decisions about whether taking the drug makes sense. And hopefully drugs to prevent or dampen ARIA will emerge, she adds.
很少有研究人员认为,对 ARIA 的担忧值得停止使用抗斑块药物,因为治疗方面的进展是阿尔茨海默氏症患者所喜闻乐见的,而且不治疗这种疾病的后果是非常严重的。此外,临床试验是在患有多种并发症的老年患者身上进行的,这些并发症可能会掩盖症状的原因。 尽管如此,许多人仍呼吁开展更多研究。"威尔科克说:"我们必须努力了解 ARIA 的潜在机制。"我们急需生物标志物来帮助我们识别风险最高的患者"。她说,最终应该可以确定每个人获得抗斑块药物帮助的几率以及受到伤害的风险,这样医生、病人和家属就可以在知情的情况下决定服用这种药物是否合理。她补充说,希望能够出现预防或抑制 ARIA 的药物。
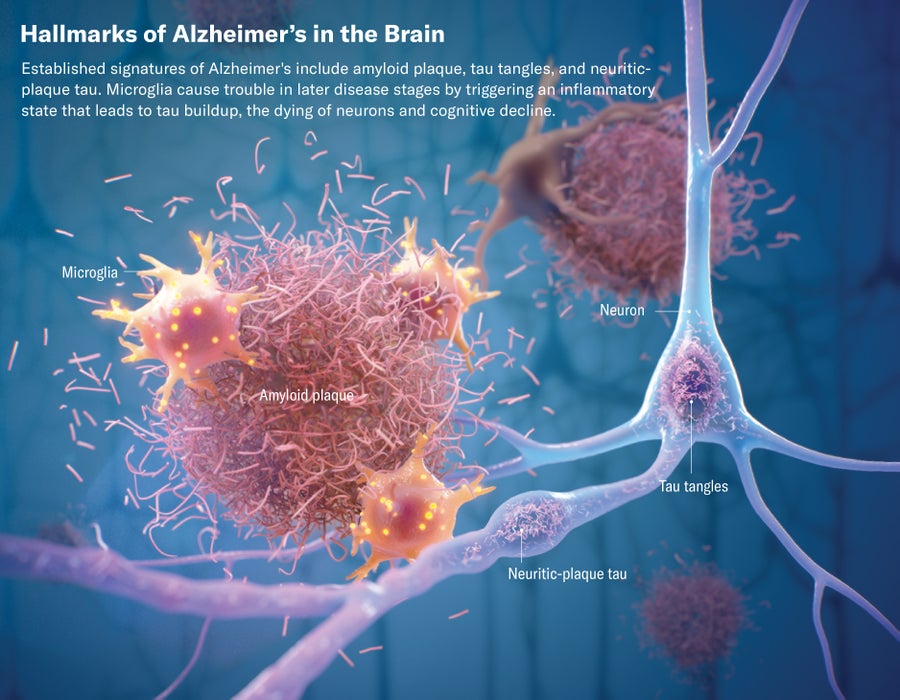
While anti-plaque strategies have been front and center when it comes to finding treatments for Alzheimer’s, another important approach has been gaining more attention recently. It involves going after the so-called tau tangles that spread through the brains of Alzheimer’s patients, particularly in the later stages.
在寻找阿尔茨海默氏症治疗方法的过程中,抗斑块策略一直是最重要的,而另一种重要方法最近也越来越受到关注。这种方法是针对在阿尔茨海默氏症患者大脑中扩散的所谓 tau 结,尤其是在晚期阶段。
Tau is a type of protein normally found within neurons, or brain cells, helping them to maintain their structure. But in Alzheimer’s disease these proteins detach, deform and stick together into tangled forms that ultimately damage the neuron. Although the exact mechanisms of Alzheimer’s remain cloudy, most researchers suspect that the buildup of plaque happens earlier in the disease, and the accumulating plaque eventually accelerates or even causes the formation and spread of tau tangles throughout the brain. Scientists used to think that amyloid and tau were competing theories but now believe they are part of the same disease cascade—that amyloid comes first and lays the groundwork for the formation of tau tangles, which is roughly when symptoms begin to emerge.
Tau 是一种通常存在于神经元或脑细胞内的蛋白质,帮助它们维持结构。但在阿尔茨海默病中,这些蛋白质会分离、变形并粘连在一起,形成纠结状,最终损害神经元。虽然阿尔茨海默氏症的确切发病机制仍不明确,但大多数研究人员怀疑,斑块的堆积发生在疾病的早期,而斑块的累积最终会加速甚至导致 tau 蛋白缠结在整个大脑中的形成和扩散。科学家们过去认为淀粉样蛋白和 tau 蛋白是相互竞争的理论,但现在认为它们是同一种疾病级联的一部分--淀粉样蛋白先出现,为 tau 蛋白缠结的形成奠定了基础,而这正是症状开始出现的大致时间。
Evidence suggests that it is the tau tangles, more than the plaque, that directly cause cognitive decline. That theory received a boost from the trial for the experimental anti-plaque drug donanemab. The trial showed that the drug did more to slow cognitive decline among patients who had plaque but did not yet have high levels of tau tangles—patients saw progression of the disease slowed by up to 60 percent.
有证据表明,直接导致认知能力下降的是头绪缠结,而不是斑块。抗斑块试验性药物多那尼单抗的试验推动了这一理论的发展。试验结果表明,这种药物在减缓斑块患者的认知能力衰退方面发挥了更大的作用--患者的疾病进展被减缓了多达 60%。
The implication of that finding is that reducing plaque can slow the accumulation of tau, but once tau has sufficiently accumulated reducing plaque alone may be not effective. “We’re already seeing a sequenced approach in drug trials, where first you try to clear out the plaque and then inject an anti-tau drug,” says Gauthier. “I suspect within two years we’ll see an effective anti-tau drug and within three years this will be the normal sequence of treatment for patients.”
这一发现的意义在于,减少斑块可以减缓tau的积累,但一旦tau积累到一定程度,单靠减少斑块可能就没有效果了。"Gauthier说:"我们已经在药物试验中看到了一种循序渐进的方法,即首先尝试清除斑块,然后注射抗tau药物。"我猜想,两年内我们就能看到有效的抗陶药物,三年内这将成为患者的正常治疗顺序。
Although plaque and tangles are the main targets of Alzheimer’s drug development efforts, other factors are known to play a role. Two of the biggest are brain inflammation and brain bleeding from damaged blood vessels. It isn’t known whether these factors become part of the disease, are risk factors for it, or are simply independent factors that exacerbate and accelerate cognitive decline.
尽管斑块和缠结是阿尔茨海默氏症药物开发工作的主要目标,但已知其他因素也在其中发挥了作用。其中最大的两个因素是脑部炎症和受损血管引起的脑出血。目前还不清楚这些因素是成为疾病的一部分,还是成为疾病的风险因素,或者仅仅是加剧和加速认知能力衰退的独立因素。
Regardless of their relationship to the disease, researchers believe attacking these problems will give Alzheimer’s patients better outcomes. That means ultimately that patients will likely be treated with a combination of drugs.
无论这些问题与疾病的关系如何,研究人员相信,解决这些问题将为阿尔茨海默病患者带来更好的治疗效果。这意味着患者最终可能会接受多种药物的综合治疗。
“In 20 years, we’ll give patients different drugs for plaque and for tangles, and maybe each will slow the decline by 25 percent or more,” says Wilcock. “Then we’ll give them something for inflammation, and maybe that will slow it by another 20 percent. Then we’ll go after the health of their blood vessels and get another 15 percent. Little by little we’ll approach the point where we can bring the cognitive decline to a full stop.”
"威尔科克说:"20 年后,我们将为患者提供不同的药物来治疗斑块和缠结,也许每种药物都能将病情恶化的速度减慢 25% 或更多。威尔科克说:"然后,我们会给他们服用一些治疗炎症的药物,也许这能再减缓 20%。然后,我们再改善他们的血管健康,再减缓 15%。渐渐地,我们就能达到完全阻止认知能力下降的目的。
Disparities 差异
Alzheimer’s puts a tremendous burden on patients, their caregivers and society at large. Patients lose years of productive life. Caregivers often sacrifice income to care for their loved ones who have the disease and their own mental health. All this has a huge impact on the world’s capacity to be productive—a trend that will only increase as populations age.
阿尔茨海默氏症给患者、照顾者和整个社会带来了巨大的负担。患者会失去多年的生产生活。照顾者往往牺牲收入来照顾患病的亲人和自己的心理健康。所有这些都对全世界的生产能力产生了巨大影响--随着人口老龄化的加剧,这种趋势只会越来越明显。
Medicine may soon be able to help by dramatically slowing or even halting the progression of Alzheimer’s. That raises concerns about how to get treatments to the people who most need them.
医学也许很快就能大幅减缓甚至阻止阿尔茨海默氏症的发展。这就引起了人们对如何为最需要的人提供治疗的关注。
One concern is cost. In the U.S., most insurance plans will cover treatments, but deductibles, co-pays and caps will typically leave patients looking at many thousands of dollars in out-of-pocket costs. Only a fraction of the estimated 1 million patients in the U.S. who could qualify for treatments are likely to be able to afford them. In addition, some groups that are more likely to get Alzheimer’s disease, including Black Americans and Hispanics, also tend to have less access to adequate healthcare and to financial resources for covering out-of-pocket costs.
一个令人担忧的问题是费用。在美国,大多数保险计划都会承保治疗费用,但免赔额、共付额和上限通常会让患者自付数千美元。 在美国约 100 万名符合治疗条件的患者中,可能只有一小部分人能够负担得起治疗费用。此外,一些更容易患阿尔茨海默病的群体,包括美国黑人和西班牙裔美国人,也往往较难获得足够的医疗保健服务和负担自付费用的经济资源。
“This is an incredibly expensive disease,” says Rabinovici, “and we’re seeing disparities for minorities in every aspect of it, from inclusion in drug trials to access to specialty care. All of us involved in research and clinical care need to think about this and issue a collective call to action.”
拉比诺维奇说:"这是一种极其昂贵的疾病","我们看到,从药物试验到获得专科护理的各个方面,少数群体都存在着差距。我们所有从事研究和临床治疗的人都需要思考这个问题,并发出集体行动的呼吁。
The U.S. healthcare system is already struggling with a shortage of clinicians, both primary care and neurologists. That’s leaving many experts worrying how the system will cope with a growing number of Alzheimer’s patients, combined with a new diagnostic and treatment protocol that will demand more time and expertise on the part of clinicians. “We’re already scheduling patients six to 12 months out in our memory clinic, and we’re turning many away,” says Jeffrey Burns, a neurologist who co-directs the Alzheimer’s Disease Research Center at the University of Kansas Medical Center. “That’s only going to get worse, especially as we develop ways to treat the disease earlier and earlier on and more patients push for those treatments.”
美国医疗保健系统已经在努力解决临床医生(包括初级保健医生和神经科医生)短缺的问题。这让许多专家担心,面对越来越多的阿尔茨海默病患者,再加上新的诊断和治疗方案需要临床医生投入更多的时间和专业知识,医疗系统将如何应对?"堪萨斯大学医学中心阿尔茨海默病研究中心的联合主任、神经科医生杰弗里-伯恩斯(Jeffrey Burns)说:"我们的记忆门诊已经在为 6 到 12 个月后的病人排期了,而且我们正在将许多病人拒之门外。"这种情况只会越来越糟,尤其是随着我们开发出越来越早治疗这种疾病的方法,越来越多的患者开始寻求这些治疗方法"。
Burns’s center has pulled together tools that help train clinicians in how to help patients more effectively and efficiently and has recruited hundreds of primary care and other doctors and nurses into a support network to provide access to the tools and facilitate information sharing. Those types of supports will help, but Burns recognizes it will be a struggle to scale them up. “The challenges are magnified by the fragmented way our healthcare system is structured,” he says. “Different hospitals and practices use different medical-record systems and have their own care protocols.”
伯恩斯的中心汇集了各种工具,帮助培训临床医生如何更有效、更高效地帮助病人,并招募了数百名初级保健医生、其他医生和护士加入支持网络,为他们提供使用工具的机会,促进信息共享。这些类型的支持将有所帮助,但伯恩斯认识到,要扩大这些支持的规模还很困难。"他说:"由于我们的医疗保健系统结构分散,挑战也就更大了。"他说:"不同的医院和诊所使用不同的医疗记录系统,并有自己的护理协议。
The U.S. public-health system, too, is fractured. The Centers for Disease Control and Prevention can issue centralized guidance, but actual public-health policy and programs are developed and implemented at the state or even local level. That disconnect will pose another obstacle to managing Alzheimer’s, asserts Heather Snyder, a molecular biologist who serves as the vice president of medical and scientific relations for the Alzheimer’s Association. That’s because coping with Alzheimer’s will require more than just better treatments for individual patients, Snyder explains. “It’s also going to take public-health actions that can impact behaviors at the population level,” she says.
美国的公共卫生系统也是支离破碎的。美国疾病控制和预防中心可以发布中央指导,但实际的公共卫生政策和计划是在州甚至地方一级制定和实施的。希瑟-斯奈德(Heather Snyder)是一位分子生物学家,她是阿尔茨海默氏症协会(Alzheimer's Association)负责医疗和科学关系的副总裁。斯奈德解释说,这是因为应对阿尔茨海默氏症需要的不仅仅是为个别患者提供更好的治疗方法。"她说:"还需要采取能够影响人群行为的公共卫生行动。
That means raising awareness of the warning signs of the disease and the need to have a conversation with a physician about them, she says. It also means finding ways to get more people to adopt healthy lifestyles and follow doctors’ advice in order to limit heart disease, obesity, diabetes, and other chronic diseases to help prevent Alzheimer’s. To help, in 2023 the Alzheimer’s Association organized a summit in Atlanta of public-health experts and officials. “We need to engage state officials in thinking about how to bring the right risk-reduction messaging to the population,” says Snyder. “There’s a lot of work to do.” To compound the challenge, preventive medicine has long been a weak point in U.S. healthcare.
她说,这意味着要提高人们对这种疾病的警示信号以及与医生进行交流的必要性的认识。这还意味着要想方设法让更多的人采用健康的生活方式,听从医生的建议,以控制心脏病、肥胖症、糖尿病和其他慢性疾病,从而帮助预防阿尔茨海默氏症。为此,阿尔茨海默氏症协会于 2023 年在亚特兰大组织了一次由公共卫生专家和官员参加的峰会。"斯奈德说:"我们需要让州政府官员参与进来,思考如何向民众传递正确的降低风险信息。"还有很多工作要做。预防医学长期以来一直是美国医疗保健的一个薄弱环节,这使得挑战更加严峻。
Issues of access are even more acute in poor nations whose healthcare systems struggle with less challenging diseases. “The kinds of healthcare resources we have in higher-income countries like the U.S. aren’t available in many countries around the world, including brain imaging and neurologists,” says Rabinovici. Not all healthcare systems around the world will make new anti-plaque and other Alzheimer’s drugs available, or cover most of its costs, experts say.
贫穷国家的医疗系统在应对难度较低的疾病时举步维艰,在这些国家,就医问题甚至更为突出。"拉比诺维奇说:"我们在美国等高收入国家拥有的医疗资源,包括脑成像和神经科医生,在世界许多国家都无法获得。专家们说,并非世界上所有的医疗保健系统都能提供新的抗斑块药物和其他阿尔茨海默氏症药物,也并非所有的医疗保健系统都能承担大部分费用。
Some pilot programs are addressing various ideas of getting healthcare to Alzheimer’s patients. In Scotland, a pilot program funded by the Davos Alzheimer’s Collaborative (DAC) is beginning to include blood tests and digital-cognitive tests into the regular checkups and national health systems. In Armenia, as part of another DAC-funded program, health workers who go out in vans to administer routine checkups will soon have blood tests and digital assessments.
一些试点项目正在探讨为阿尔茨海默病患者提供医疗保健的各种想法。在苏格兰,一项由达沃斯阿尔茨海默氏症合作组织(DAC)资助的试点项目开始将血液检测和数字认知测试纳入常规体检和国家医疗系统。在亚美尼亚,作为达沃斯老年痴呆症合作组织(DAC)资助的另一项计划的一部分,乘车外出进行例行体检的医务工作者不久将进行血液检测和数字评估。
In spite of these many concerns, the overall feeling in the field is that things are finally moving fast in the right direction. “This is an incredibly hopeful and exciting time,” says Wilcock. “For the 24 years I’ve been in the field we’ve been telling patients that treatments were five years away. Now we can finally say we’re here.”
尽管存在诸多顾虑,但该领域的总体感觉是,事情终于朝着正确的方向快速发展。"威尔科克说:"这是一个充满希望和令人兴奋的时刻。"在我从业的 24 年里,我们一直告诉病人,治疗还需要五年时间。现在,我们终于可以说我们到了。
This article is part of The New Age of Alzheimer’s, a special report on the advances fueling hope for ending this devastating disease.
这篇文章是《阿尔茨海默氏症的新时代》(The New Age of Alzheimer's )的一部分,该特别报道介绍了为终结这种毁灭性疾病而取得的进展。




